Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124


Quick Answer: Gut health refers to the balance and function of the gastrointestinal system, especially the gut microbiome — the trillions of bacteria, fungi, and other microorganisms living in your digestive tract. Poor gut health causes symptoms like bloating, fatigue, brain fog, and weakened immunity. You can improve gut health naturally by eating more fiber and fermented foods, reducing ultra-processed foods, managing stress, sleeping 7–9 hours, and taking evidence-based probiotics.
You have probably heard the phrase ‘trust your gut.’ It turns out, science agrees — more literally than most people realize.
Your gut is not just a digestive organ. It is an intricate ecosystem of trillions of microorganisms, a central hub of your immune system, and a communication center that talks directly to your brain. When your gut is thriving, everything works better: your energy, your mood, your immunity, your metabolism, even the clarity of your thinking.
When your gut is struggling — disrupted by a poor diet, chronic stress, antibiotics, or a sedentary lifestyle — the consequences extend far beyond digestion. Fatigue, anxiety, skin conditions, autoimmune flare-ups, weight gain, and cognitive fog are all increasingly linked to gut dysfunction.
This guide gives you everything you need to understand gut health, recognize the warning signs of a struggling gut, identify the root causes of gut dysfunction, and follow a clear, evidence-based plan to improve your gut health naturally — starting today.
Why this matters: According to the World Health Organization (WHO), chronic non-communicable diseases — most of which have a gut health component — account for 74% of all global deaths. The gut microbiome is now at the center of some of the most important medical research of the 21st century.
Gut health refers to the overall function, balance, and integrity of your gastrointestinal (GI) system — from your esophagus to your large intestine — and specifically to the health of the gut microbiome: the vast community of microorganisms living within it.
A healthy gut is characterized by:
Gut health is not simply the absence of digestive symptoms. It is a systemic health state — because what happens in your gut does not stay in your gut. The gut microbiome influences virtually every organ system in the body.
| Function | What It Does | What Goes Wrong Without It |
| Digestion | Breaks down food into absorbable nutrients | Malnutrition, deficiencies |
| Absorption | Transfers nutrients into the bloodstream | Low energy, anaemia, bone loss |
| Immune regulation | 70-80% of immune cells live here; trains immune tolerance | Autoimmune disease, allergies, infection |
| Gut-brain signaling | Produces 90% of serotonin; communicates via vagus nerve | Anxiety, depression, brain fog |
| Barrier function | Prevents toxins and bacteria from entering the bloodstream | Leaky gut, systemic inflammation |
Key Stat: Your gastrointestinal tract contains approximately 38 trillion microorganisms — more cells than the rest of your entire body combined, according to research published in Cell.
The gut microbiome is the collective community of bacteria, viruses, fungi, and other microorganisms living in your gastrointestinal tract — predominantly in the large intestine (colon). It contains over 1,000 different bacterial species, encoding more than 3 million genes — roughly 150 times the number in the human genome.
This microbial community is not a passive passenger in your body. It is an active metabolic organ that:
| Bacterium / Group | Why It Matters for Your Health |
| Lactobacillus | Produces lactic acid; supports vaginal and gut immunity; reduces anxiety (GABA production) |
| Bifidobacterium | Produces B vitamins; reduces inflammation; critical for infant immune development |
| Faecalibacterium prausnitzii | Largest butyrate producer; powerfully anti-inflammatory; depleted in IBD, depression, obesity |
| Akkermansia muciniphila | Maintains gut mucosal barrier; associated with leanness, reduced diabetes risk, better immunotherapy response |
| Roseburia intestinalis | Butyrate producer; improves insulin sensitivity; depleted in type 2 diabetes |
| Christensenellaceae | Associated with longevity and leanness; highly heritable |
| Bacteroides fragilis | Supports immune tolerance and anti-inflammatory signalling |
| Enterobacteriaceae (excess) | Pro-inflammatory when overgrown; associated with leaky gut and metabolic disease |
The most consistently validated marker of a healthy gut microbiome is diversity — the number and variety of different microbial species present. Greater diversity correlates with:
The American Gut Project — the world’s largest citizen science microbiome study, published in mSystems — found that people who eat 30 or more different plant foods per week have dramatically greater microbiome diversity than those eating fewer than 10. This single finding is one of the most actionable in all of nutritional science.
Actionable Takeaway: Aim to eat 30+ different plant foods per week. This includes vegetables, fruits, legumes, whole grains, nuts, seeds, herbs, and spices — each variety counts separately.
Poor gut health — a state called gut dysbiosis — rarely announces itself with a single, obvious symptom. It typically manifests as a cluster of seemingly unrelated issues that together paint a clear picture of a disrupted gut-body ecosystem.
Here are the 10 most important warning signs, organized from most to least recognized:
Persistent bloating, gas, diarrhea, constipation, or alternating bowel habits are the most direct indicators of gut health problems. These symptoms reflect an imbalanced microbiome, impaired gut motility, or an inflammatory process in the gut lining. According to the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK), irritable bowel syndrome (IBS) — a gut dysbiosis-driven condition — affects an estimated 10–15% of adults globally.
Important: frequent bloating after eating, urgency, or significant discomfort that disrupts daily life should be evaluated by a gastroenterologist to rule out conditions like IBD, celiac disease, or SIBO.
If you consistently feel exhausted despite adequate sleep, your gut may be the culprit. The gut microbiome synthesizes B vitamins essential for energy metabolism. A dysbiotic gut also reduces iron and magnesium absorption — two of the most common nutritional drivers of fatigue. Additionally, gut-derived LPS (bacterial endotoxins) that leak through a compromised gut lining trigger systemic inflammation — one of the most potent causes of medically unexplained fatigue.
The gut produces approximately 90% of the body’s serotonin and significant quantities of dopamine precursors. Gut dysbiosis disrupts this production, impairing mood, motivation, working memory, and mental clarity. Gut-derived neuroinflammation — where LPS crosses the blood-brain barrier — directly impairs cognitive function. Research from the NIH confirms altered tryptophan metabolism (the serotonin precursor pathway) is a consistent finding in both gut dysbiosis and cognitive impairment.
The gut microbiome regulates metabolism, appetite hormones (GLP-1, PYY, ghrelin), and energy extraction from food. Dysbiosis characterized by high Firmicutes and low Bacteroidetes ratios is associated with greater caloric extraction and obesity. Conversely, gut-driven malabsorption can cause unexplained weight loss. Both weight gain and weight loss without dietary change can signal a gut microbiome imbalance.
The gut-skin axis is a well-established bidirectional relationship. Gut dysbiosis produces systemic inflammation and immune dysregulation that manifests in the skin. Research confirms that people with psoriasis, atopic dermatitis, rosacea, and acne show consistent and specific gut microbiome differences from healthy controls. Elevated gut-derived LPS activates Toll-like receptors in skin, driving the inflammatory cascades underlying these conditions.
Seventy to eighty percent of your immune cells reside in the gut-associated lymphoid tissue (GALT). A healthy, diverse microbiome trains these cells to distinguish threats from harmless antigens. Gut dysbiosis impairs this immune education — resulting in both overreactivity (allergies, autoimmunity) and underreactivity (susceptibility to infections). Frequent colds, slow wound healing, or recurring UTIs and thrush all suggest compromised gut-immune function.
The gut-brain axis means that gut health is mental health. Gut bacteria produce GABA (calming), serotonin (mood-regulating), and dopamine precursors. A 2021 review in General Psychiatry found that both probiotic supplementation and dietary gut health interventions significantly reduced anxiety and depression scores. Conversely, gut-derived systemic inflammation is increasingly recognized as a driver of major depressive disorder — with anti-inflammatory interventions showing antidepressant effects in clinical trials.
Developing new food sensitivities — particularly to foods you previously tolerated well — is a classic sign of compromised gut barrier integrity (leaky gut). When tight junctions between intestinal cells loosen, partially digested food proteins enter the bloodstream, triggering immune responses. Over time, this creates the pattern of expanding food intolerances, histamine sensitivity, and reactivity to seemingly random foods that many patients with gut dysfunction experience.
The research linking gut dysbiosis to autoimmune disease is now robust. Conditions including rheumatoid arthritis, type 1 diabetes, multiple sclerosis, lupus, Hashimoto’s thyroiditis, and psoriatic arthritis all show consistent gut microbiome abnormalities. The mechanism: leaky gut allows bacterial antigens into systemic circulation, where molecular mimicry (bacterial proteins resembling self-tissue proteins) can trigger the immune system to attack the body’s own cells.
The gut microbiome follows a circadian rhythm that directly influences your sleep-wake cycle. Gut bacteria produce melatonin precursors and regulate the HPA axis (stress response) that governs sleep quality. NIH research confirms that gut dysbiosis — even without clinically obvious digestive symptoms — disrupts sleep architecture and reduces deep sleep duration. Conversely, poor sleep devastates microbiome diversity within 2–3 days, creating a self-reinforcing gut-sleep dysfunction cycle.
| Symptom | Most Likely Gut Mechanism |
| Bloating / gas | Dysbiosis, SIBO, impaired gut motility |
| Fatigue | B vitamin deficiency, LPS-driven inflammation, poor iron/Mg absorption |
| Brain fog | Reduced serotonin, gut-derived neuroinflammation, LPS crossing blood-brain barrier |
| Skin flares | Gut-skin axis: LPS-driven dermal inflammation, Th2 immune skewing |
| Anxiety / low mood | Reduced GABA and serotonin production, vagal dysregulation |
| Food intolerances | Leaky gut — undigested proteins triggering systemic IgG/IgE responses |
| Frequent infections | GALT dysfunction — impaired mucosal IgA and innate immunity |
| Autoimmune flares | Molecular mimicry from gut-derived antigens crossing compromised barrier |
| Weight changes | Altered Firmicutes/Bacteroidetes ratio, disrupted GLP-1 and ghrelin signalling |
| Poor sleep | Disrupted circadian microbiome rhythm, reduced melatonin precursors |
Important: If you experience several of these symptoms simultaneously, consult your healthcare provider. Many have overlapping causes that warrant investigation — including stool microbiome analysis, intestinal permeability testing, and relevant blood panels.
Understanding the causes of gut dysbiosis is the foundation of any meaningful gut health intervention. Here are the most evidence-based drivers:
The modern Western diet — high in ultra-processed foods (UPFs), refined sugars, seed oils, emulsifiers, and artificial sweeteners — is the single most damaging force acting on the gut microbiome. Research in Nature found that common food emulsifiers (carboxymethylcellulose and polysorbate-80) erode the gut’s protective mucus layer and increase intestinal permeability at concentrations routinely present in processed foods. Another landmark Nature paper demonstrated that artificial sweeteners (sucralose, aspartame, saccharin) significantly disrupt gut microbiome composition and induce glucose intolerance.
The problem: UPFs now account for more than 60% of caloric intake in the US and UK. They displace the fiber-rich plant foods that beneficial gut bacteria need to survive.
Dietary fiber is the primary fuel source for SCFA-producing gut bacteria. Without it, these bacteria — including critical species like Faecalibacterium prausnitzii and Roseburia intestinalis — starve and die. Research from Stanford University showed that a low-fiber diet produces rapid and measurable depletion of beneficial bacterial species within days. The WHO recommends at least 25g of fiber daily; most Western adults consume under 15g.
Antibiotics are life-saving medicines — but they are profoundly non-selective. A single course of broad-spectrum antibiotics can eliminate up to 30% of gut bacterial diversity, with some species taking months or years to recover. Research published in mBio found that microbiome disruption from a single antibiotic course is detectable for up to 12 months. In some individuals — particularly those who received antibiotics in early childhood — certain species may never fully recover. The CDC estimates that 30% of antibiotic prescriptions in the US are unnecessary.
The gut-brain axis operates in both directions — which means that stress damages the gut just as powerfully as gut dysfunction causes mental distress. Cortisol (the primary stress hormone) directly increases intestinal permeability, reduces beneficial Lactobacillus populations, and stimulates overgrowth of pathogenic bacteria. Research from the American Psychological Association (APA) confirms that perceived psychological stress is a significant predictor of gut microbiome dysbiosis, IBS flares, and IBD relapse.
The gut microbiome is governed by a circadian rhythm synchronized to your sleep-wake cycle. Shift work, chronic sleep deprivation, and irregular sleep schedules disrupt this rhythm, rapidly reducing Bifidobacterium populations and increasing pro-inflammatory species. Just two days of sleep restriction produce measurable gut dysbiosis in controlled studies.
Regular exercise is a significant driver of microbiome diversity. A 2019 review in Gut Microbes found that exercise promotes the growth of butyrate-producing bacteria and reduces pro-inflammatory Proteobacteria. Sedentary individuals consistently show lower microbiome diversity than active peers, independent of dietary differences.
Alcohol is directly toxic to the gut mucosal lining. Even moderate consumption reduces Bacteroidetes populations, increases Proteobacteria, and significantly increases intestinal permeability. Heavy alcohol use is associated with severe gut dysbiosis, bacterial overgrowth, and elevated circulating LPS — a pattern called ‘alcoholic leaky gut syndrome’ that precedes alcoholic liver disease.
Multiple commonly prescribed medications significantly disrupt the gut microbiome:
How your microbiome was established in early life has lasting consequences. Factors that reduce initial microbial colonization include caesarean birth (bypasses vaginal microbiome seeding), formula feeding (breast milk contains HMOs — human milk oligosaccharides that specifically feed Bifidobacterium), antibiotic use in the first 3 years of life, and high-sanitization environments. Research from the NIH confirms these early-life factors are among the strongest predictors of adult microbiome diversity and allergy/autoimmune disease risk.
Pesticides, heavy metals, microplastics, chlorinated water, and air pollution all exert measurable negative effects on gut microbiome composition. Glyphosate — the world’s most widely used herbicide — inhibits the shikimate pathway used by many beneficial gut bacteria. Research in the journal Toxicology found detectable microbiome disruption at glyphosate exposure levels considered safe by regulators.
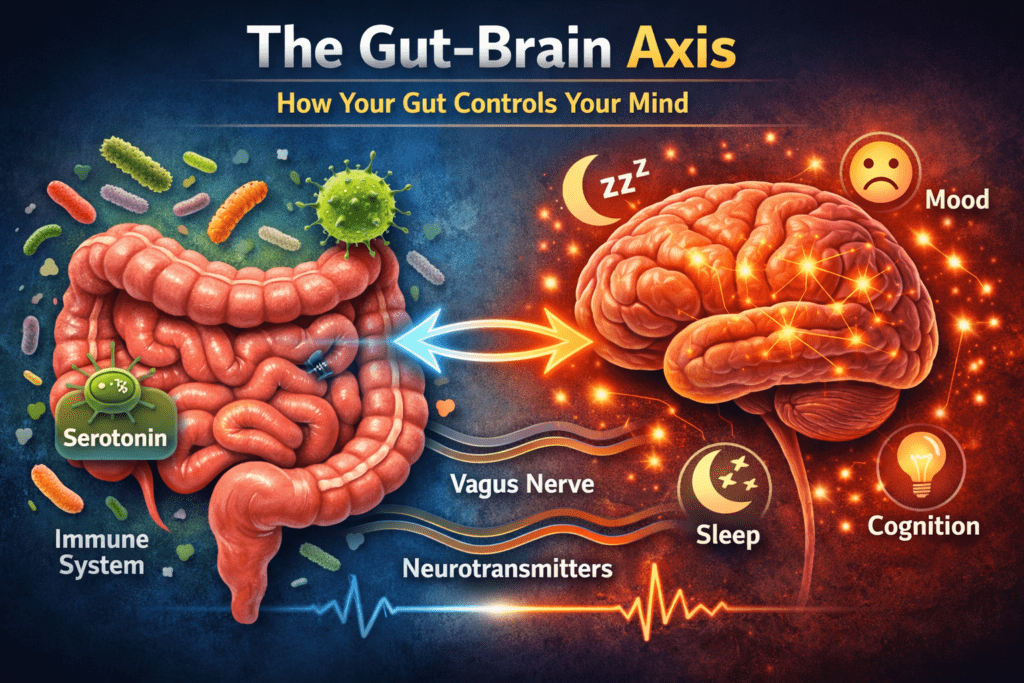
The gut-brain axis is the bidirectional communication network connecting your gastrointestinal system and your central nervous system. It operates through four primary pathways:
The vagus nerve is the longest nerve in the body, running from the brainstem directly to the gut. Approximately 80–90% of its fibers carry information FROM the gut TO the brain — meaning your gut is continuously reporting on its microbial status, inflammatory state, and nutrient content to your brain. A healthy, diverse microbiome sends calming, regulatory signals. A dysbiotic gut sends chronic stress signals — keeping the nervous system in a state of low-level alert that manifests as generalized anxiety, hypervigilance, and reduced stress resilience.
Your gut contains approximately 500 million neurons — more than your spinal cord. This enteric nervous system (ENS) operates largely independently, governing gut motility, secretion, and blood flow. It communicates bidirectionally with the brain, and disruption of the ENS is now understood to contribute to conditions ranging from IBS to Parkinson’s disease (which is increasingly believed to originate in the ENS).
Your gut bacteria produce or directly regulate the production of every major neurotransmitter:
Dysbiosis depletes the bacteria that produce these compounds — directly contributing to anxiety, depression, attention difficulties, and motivation disorders.
Gut-derived LPS (lipopolysaccharides from the outer membrane of gram-negative bacteria) leaks through a compromised gut barrier into systemic circulation, where it crosses the blood-brain barrier and activates resident brain immune cells (microglia). Chronic microglial activation produces neuroinflammation — which is now recognized as a core feature of depression, anxiety, Alzheimer’s disease, and Parkinson’s disease.
Research Highlight: A 2021 study in Cell found that a high-fermented-food diet reduced 19 key inflammatory proteins — several directly linked to depression, dementia, and autoimmune disease — more effectively than a high-fiber diet alone over 17 weeks.
This is not a list of vague tips. This is a specific, sequenced protocol based on the strongest evidence available — ordered to produce the fastest, most durable gut health improvements.
Before rebuilding, you must stop the active damage. For the first week, eliminate or dramatically reduce:
What to expect in week 1: some digestive adjustment, possible temporary increase in cravings, gradual reduction in bloating.
Dietary fiber is the single most impactful intervention for gut microbiome diversity. It feeds the SCFA-producing bacteria that reduce inflammation, regulate immunity, and produce protective butyrate. The target is 35–40g daily, reached gradually to avoid bloating.
The most gut-beneficial fiber sources (ranked by evidence):
Pro Tip: Cook and cool your rice, potatoes, and pasta before eating them. Cooling converts some digestible starch into resistant starch — a powerful prebiotic. Reheat gently — the resistant starch largely survives reheating.
Fermented foods introduce live beneficial bacteria directly into your gut while also providing organic acids, enzymes, and bioactive compounds that support gut barrier integrity. A Stanford University clinical trial found that a high-fermented food diet increased microbiome diversity and reduced 19 inflammatory proteins more effectively than a high-fiber diet alone.
The hierarchy of fermented foods for gut health (best to good):
Start with 1–2 tablespoons daily and increase gradually over 2–3 weeks to allow your gut to adjust.
Once dietary foundations are established, targeted supplements fill gaps and accelerate gut healing:
Look for: multi-strain formulas with at least 10 billion CFU (colony-forming units). Key strains backed by the strongest evidence:
Cycle probiotic strains every 8–12 weeks to maintain diversity rather than dominance of one strain.
If dietary fiber is insufficient, prebiotic supplements directly feed beneficial bacteria:
Glutamine is the primary fuel source for enterocytes (gut lining cells) and is essential for maintaining tight junctions. Dose: 5–10g daily in the morning on an empty stomach. NIH-indexed research confirms glutamine supplementation reduces intestinal permeability.
Specifically shown to repair gut mucosal damage. Dose: 75mg twice daily with food. Particularly valuable for individuals recovering from NSAID use, infections, or inflammatory bowel conditions.
The gut’s circadian rhythm means that sleep is when the microbiome rebalances and the gut lining repairs. Inadequate sleep devastates this process. The CDC recommends 7–9 hours for adults. For optimal gut health:
Regular aerobic exercise is one of the most powerful, drug-free interventions for increasing gut microbiome diversity. A landmark study in the journal Gut found that rugby players had significantly more diverse microbiomes than sedentary controls — even after adjusting for diet.
The microbiome-exercise sweet spot:
Note: Excessive endurance exercise (ultra-marathon level) can temporarily increase gut permeability during and immediately after training — a known phenomenon called ‘exercise-induced gut leakage’. Recovery nutrition and hydration are critical.
No dietary or supplement protocol can fully compensate for the gut-damaging effects of chronic psychological stress. Cortisol directly increases intestinal permeability, reduces Lactobacillus populations, and activates the HPA axis in a self-reinforcing cycle that perpetuates both gut dysfunction and anxiety.
The three most evidence-based stress-gut interventions:
The 7-Step Protocol Summary: Remove disruptors → Build fiber → Add fermented foods → Supplement strategically → Optimise sleep → Exercise consistently → Manage stress. Follow this sequence for 8–12 weeks for measurable gut health transformation.
The best diet for gut health is one that maximises plant diversity, prioritises fermented foods, and minimises gut-disrupting processed ingredients. Here is the science-ranked list of the most gut-beneficial foods:
| Food | Key Gut Health Benefit |
| Lentils & legumes | Highest prebiotic density; increase Faecalibacterium prausnitzii and Roseburia |
| Kefir (plain, unsweetened) | Up to 61 microbial strains; superior probiotic diversity |
| Kimchi | Lactobacillus plantarum; prebiotic fiber; anti-inflammatory polyphenols |
| Oats (rolled/steel-cut) | Beta-glucan: feeds Bifidobacterium; reduces LDL; lowers post-meal glucose |
| Blueberries & dark berries | Anthocyanins increase Akkermansia muciniphila; reduce gut inflammation |
| Garlic & onions | Inulin and FOS — top prebiotic fiber sources; antimicrobial against pathogens |
| Sauerkraut (live) | Lactobacillus species; supports gut barrier; anti-inflammatory |
| Fatty fish (salmon, sardines) | Omega-3s reduce gut-derived LPS response; anti-inflammatory microbiome shift |
| Extra-virgin olive oil | Polyphenols increase Bifidobacterium and Lactobacillus; reduces gut inflammation |
| Dark chocolate (85%+) | Cacao polyphenols act as prebiotics; increase Bifidobacterium by up to 40% |
| Broccoli & cruciferous veg | Sulforaphane and indole-3-carbinol; support gut mucosal immunity; prebiotic fiber |
| Bone broth | Glutamine and collagen for tight junction repair; reduces leaky gut |
| Green banana / cooled potato | High resistant starch feeds butyrate-producing bacteria |
| Asparagus & Jerusalem artichoke | Highest inulin content of any vegetable; powerful Bifidobacterium feeders |
| Plain full-fat yogurt | Live Lactobacillus and Streptococcus thermophilus; protein; calcium |
Knowing what to avoid is as important as knowing what to eat. These foods have the strongest evidence for gut microbiome harm:
| Food / Ingredient | How It Harms the Gut |
| Ultra-processed foods (UPFs) | Emulsifiers erode mucus layer; additives disrupt microbiome; displace fiber-rich foods |
| Artificial sweeteners | Alter microbiome composition; impair glucose tolerance (Nature, 2014) |
| Refined sugar (especially HFCS) | Feeds Candida and pathogenic Enterobacteriaceae; reduces Bacteroidetes |
| Refined seed oils (in excess) | Omega-6 excess shifts microbiome to pro-inflammatory composition |
| Alcohol (especially spirits/beer) | Directly toxic to gut lining; increases intestinal permeability; reduces Bacteroidetes |
| Red/processed meat (excess) | Drives TMAO production; reduces microbial diversity; pro-inflammatory gut shift |
| Emulsifiers (polysorbate-80 etc) | Physically erode protective gut mucus layer; trigger low-grade gut inflammation |
| Carrageenan | Common thickener in ‘healthy’ foods; linked to gut inflammation in multiple studies |
| Fried foods | Oxidized fats damage gut epithelium; high in gut-disrupting additives |
| Excessive caffeine | Stimulates HPA axis; increases gut motility abnormally; may worsen leaky gut |
Note on gluten: Gluten does not harm the gut for most people. However, individuals with celiac disease (approximately 1% of the population) and non-celiac gluten sensitivity (estimated 6%) experience significant gut damage from gluten. If you suspect gluten is problematic for you, consult your doctor before going gluten-free — testing accuracy requires gluten to be in your diet.
Probiotics are one of the most researched — and most misunderstood — supplements in the health industry. Here is the honest, evidence-based picture:
Refrigerated vs shelf-stable: both can be effective — stability depends on the specific strains and encapsulation, not refrigeration per se.
Bottom line on probiotics: They are most effective when used alongside a high-fiber, fermented-food-rich diet — not as a replacement for it. Think of them as a direct bacterial top-up that works best when the dietary environment supports the bacteria you are adding.
This is one of the most common questions in gut health — and the honest answer is: it depends. The timeline varies based on the severity of dysbiosis, the specific gut condition, your starting dietary baseline, consistency of the intervention, and underlying factors like medication use and stress levels.
That said, research provides useful general timelines:
| Timeframe | What to Expect |
| Days 1–7 | Reduced bloating as gut-disrupting foods are removed; possible temporary adjustment symptoms (increased gas as microbiome shifts) |
| Weeks 1–2 | Measurable microbiome composition changes detectable within 2 weeks of dietary change (Stanford research). Improved energy and mood often noticed first. |
| Weeks 2–4 | Gut motility improvements; reduced food sensitivity reactions as intestinal permeability begins to improve; sleep quality often improves |
| Weeks 4–8 | Significant reductions in digestive symptoms (IBS studies show peak benefit at 4–8 weeks); skin conditions often begin improving via the gut-skin axis |
| Weeks 8–12 | Measurable improvements in systemic inflammatory markers; HbA1c improvements in prediabetic individuals; anxiety and mood scores improve in gut-brain axis studies |
| Months 3–6 | Establishment of new microbiome baseline; sustainable improvements in energy, cognition, and immune resilience; gut lining structural repair (collagen remodelling) |
| 6–12 months | Full microbiome diversity establishment in previously severely depleted individuals; autoimmune marker improvements in some studies |
Important nuance: microbiome changes happen fast (within days to weeks) but structural gut lining repair takes longer. Symptom improvement often precedes full microbiome rehabilitation. Do not interpret lack of immediate symptom relief as failure — the underlying biology is changing even when symptoms lag.
Research Note: A study in Nature found measurable gut microbiome composition changes within 3–4 days of dietary change. However, the same research confirmed that short-term dietary changes produce only temporary microbiome shifts — sustained change requires sustained dietary habits.
Not all supplements are equal. Here is an evidence-ranked guide to the most studied gut health supplements:
| Supplement | Evidence Level | Best For |
| Multi-strain probiotic | High | IBS, antibiotic recovery, immune support, anxiety |
| Psyllium husk | High | IBS-C, cholesterol, blood sugar regulation |
| L-Glutamine | Moderate-High | Leaky gut repair, post-infection recovery, IBD support |
| Zinc carnosine | Moderate-High | Gut mucosal healing, NSAID-related gut damage |
| Magnesium glycinate | Moderate-High | Gut motility, sleep, stress reduction |
| Omega-3 (EPA+DHA) | High | Gut inflammation reduction, microbiome anti-inflammatory shift |
| Vitamin D3 + K2 | Moderate-High | Gut mucosal immunity, barrier integrity, microbiome regulation |
| Berberine | Moderate | Blood sugar regulation, Akkermansia muciniphila increase |
| Collagen peptides | Moderate | Gut lining structural support (glycine + proline) |
| Digestive enzymes | Moderate | Immediate symptom relief in hypochlorhydria or enzyme insufficiency |
| Curcumin (with piperine) | Moderate | Gut inflammation, IBD/IBS symptom reduction |
| Slippery elm bark | Low-Moderate | Soothing inflamed gut mucosa — traditional use, limited RCTs |
| Deglycyrrhizinated licorice (DGL) | Low-Moderate | Acid reflux, gastric mucosal support |
Disclaimer: Always consult your healthcare provider before starting gut health supplements, particularly if you are pregnant, have a chronic health condition, or take prescription medications. Supplements are not a replacement for medical treatment.
Gut health refers to how well your digestive system functions and how balanced your gut microbiome is. A healthy gut digests food efficiently, absorbs nutrients properly, maintains a strong intestinal barrier, and supports a well-functioning immune system and brain.
The 10 most common signs are: chronic bloating/gas/constipation/diarrhea; persistent fatigue; brain fog; unexplained weight changes; skin conditions (acne, eczema, psoriasis); frequent illness; anxiety or depression; food intolerances; autoimmune flares; and poor sleep quality.
Gut dysbiosis is an imbalance in the gut microbiome — specifically a loss of beneficial bacterial diversity and/or overgrowth of harmful species. It is the underlying state that drives most gut health-related symptoms and conditions.
Yes — directly and profoundly. The gut-brain axis means the gut produces approximately 90% of the body’s serotonin, significant quantities of GABA (the calming neurotransmitter), and communicates with the brain via the vagus nerve. Gut dysbiosis is increasingly linked to anxiety, depression, brain fog, and cognitive decline. Multiple clinical trials show that probiotic supplementation and gut-healthy diets reduce anxiety and depression scores.
Leaky gut (intestinal hyperpermeability) is a real, measurable condition in which the tight junctions between intestinal cells loosen, allowing bacteria, toxins, and undigested food particles to enter the bloodstream. It is associated with chronic inflammation, autoimmune conditions, food sensitivities, and metabolic disease. It is not always a standalone diagnosis — it is often a feature of underlying conditions including IBD, celiac disease, and severe dysbiosis.
Follow the 7-step protocol in this guide: (1) Remove gut disruptors — ultra-processed foods, alcohol, artificial sweeteners; (2) Build the fiber foundation to 35–40g daily; (3) Add fermented foods daily; (4) Supplement strategically with probiotics, L-glutamine, and zinc carnosine; (5) Optimise sleep to 7–9 hours; (6) Exercise 150+ minutes per week; (7) Manage stress through mindfulness and breathing practices.
Gut microbiome composition changes within 2–4 weeks of dietary change. Digestive symptoms often improve within 4–8 weeks. Full microbiome restoration after significant dysbiosis can take 3–12 months of consistent dietary and lifestyle changes.
Yes — for specific conditions, the evidence is strong. Probiotics reduce antibiotic-associated diarrhea, IBS symptoms, anxiety, and eczema risk (in infants). Choose multi-strain formulas with strain-specific evidence, minimum 10 billion CFU, and third-party testing. Probiotics are most effective alongside a high-fiber diet.
The most evidence-backed dietary patterns for gut health are: the Mediterranean diet (highest diversity score and best inflammatory marker outcomes), the MIND diet (best for gut-brain health), and a whole-food, plant-diverse diet that includes fermented foods and 30+ different plant foods per week. All three share a common foundation: high fiber, minimal processing, abundant polyphenols, and regular fermented food inclusion.
Yes — significantly. Cortisol directly increases intestinal permeability, reduces Lactobacillus populations, and alters gut motility. Chronic stress is a confirmed trigger for IBS flares and IBD relapses. Managing stress is not optional in gut health — it is as important as diet.
This pillar article is the foundation of the SolidHealthInfo Gut Health content hub. For deeper dives into specific topics covered here, explore these supporting articles:
All claims in this article are supported by peer-reviewed research or official health authority guidance. Primary sources:
WHO — Healthy Diet: https://www.who.int/news-room/fact-sheets/detail/healthy-diet
NIH — National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK): https://www.niddk.nih.gov/health-information/digestive-diseases
CDC — Physical Activity Guidelines: https://www.cdc.gov/physicalactivity/basics/adults/index.htm
CDC — Sleep Recommendations: https://www.cdc.gov/sleep/about_sleep/how_much_sleep.html
NIH — Office of Dietary Supplements: https://ods.od.nih.gov/
NIH — National Institute on Aging, Microbiome: https://www.nia.nih.gov/research/dn/microbiome-aging
NIH — National Center for Complementary and Integrative Health: https://www.nccih.nih.gov/
American Gut Project (mSystems, 2018) — 30 plant foods/week and microbiome diversity: https://msystems.asm.org/content/3/3/e00031-18
Stanford — Fermented foods vs fiber (Cell, 2021): https://www.cell.com/cell/fulltext/S0092-8674(21)00754-6
Nature — Artificial sweeteners and microbiome: https://www.nature.com/articles/nature13793
NIH — Gut serotonin production (PMC7238917): https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7238917/
NIH — Glutamine and intestinal permeability (PMC4369670): https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4369670/
General Psychiatry — Probiotics reduce anxiety (PMC7001367): https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7001367/
NIH — Microbiome and Alzheimer’s (PMC7235093): https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7235093/
Weizmann Institute — Personalised glucose response and microbiome (PMC4681099): https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4681099/
Nature — Cleveland Clinic TMAO and cardiovascular risk (PMC4905595): https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4905595/Medical Disclaimer: The information in this article is for educational purposes only and does not constitute medical advice, diagnosis, or treatment. Gut health symptoms can have serious underlying causes. Always consult a qualified gastroenterologist or healthcare provider before making changes to your diet, supplement routine, or treatment plan — especially if you have been diagnosed with a digestive disorder, autoimmune condition, or chronic disease.